Spotfire® for Life Sciences
Confident decisions for life sciences manufacturing
Unify quality, process, and production in one decision layer.

From fragmented quality data to controlled, compliant operations.
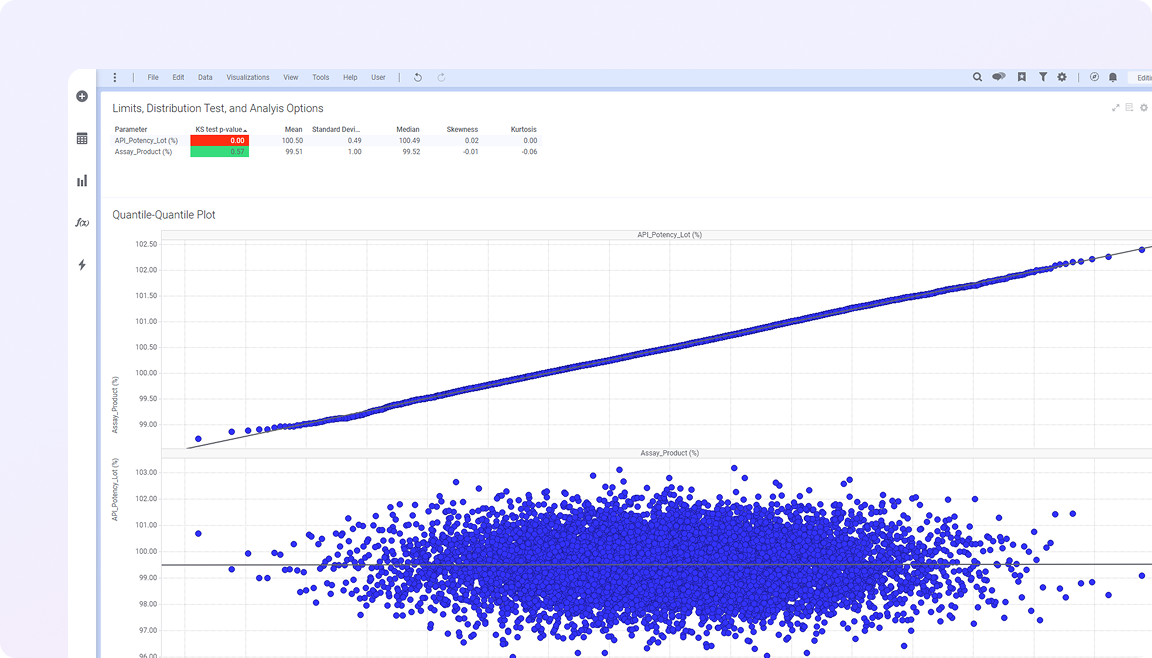
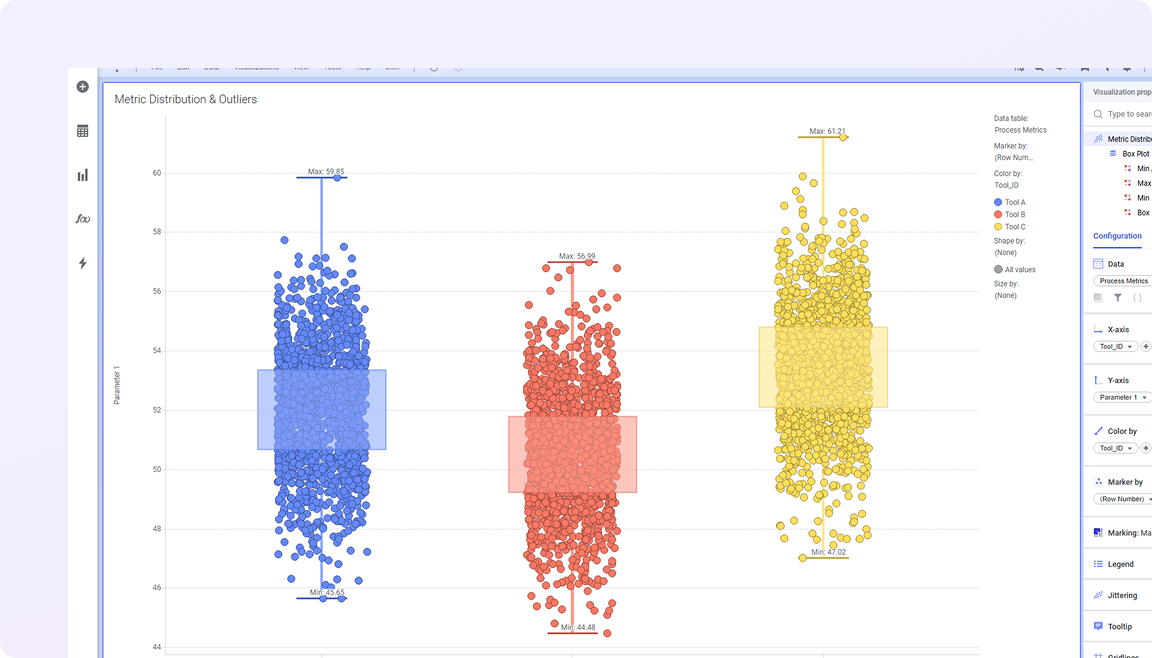
Life sciences manufacturing data spans batch records, process parameters, equipment performance, and quality testing, often across validated and disconnected systems. This fragmentation makes it difficult to understand process behavior, maintain traceability, and identify quality risks early.
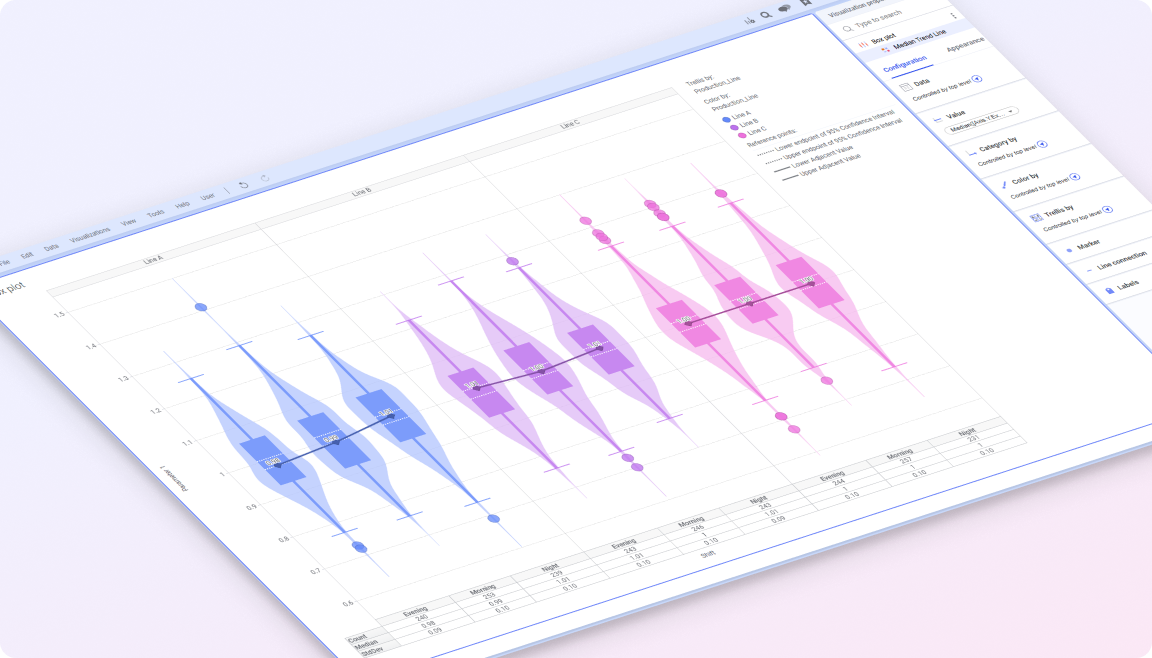
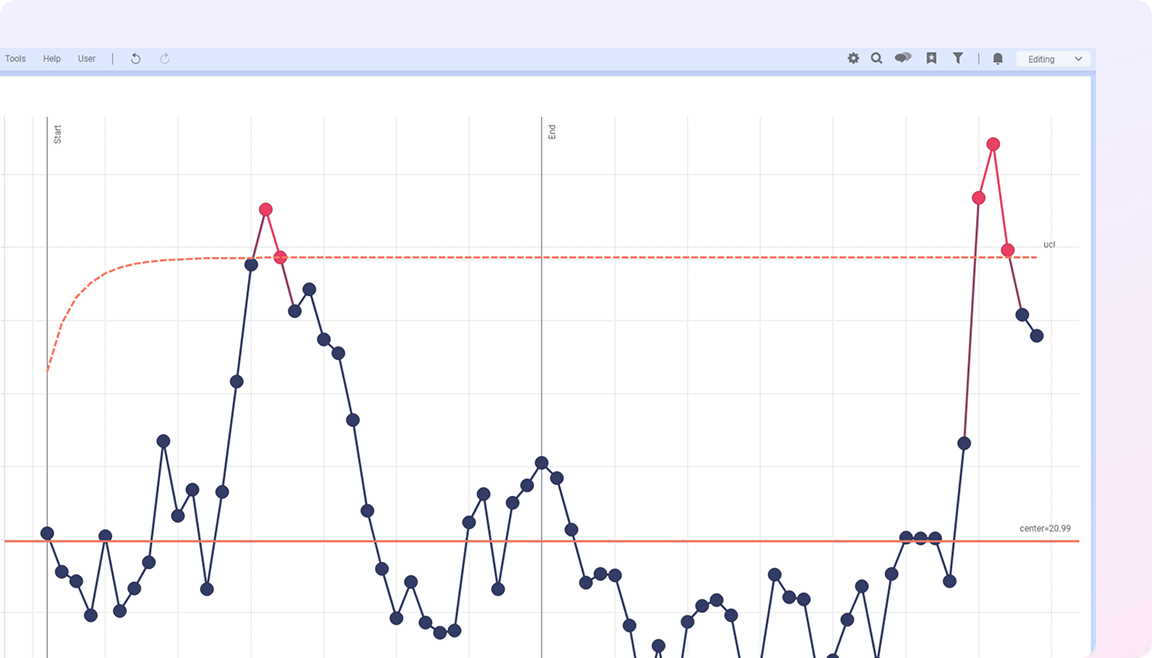
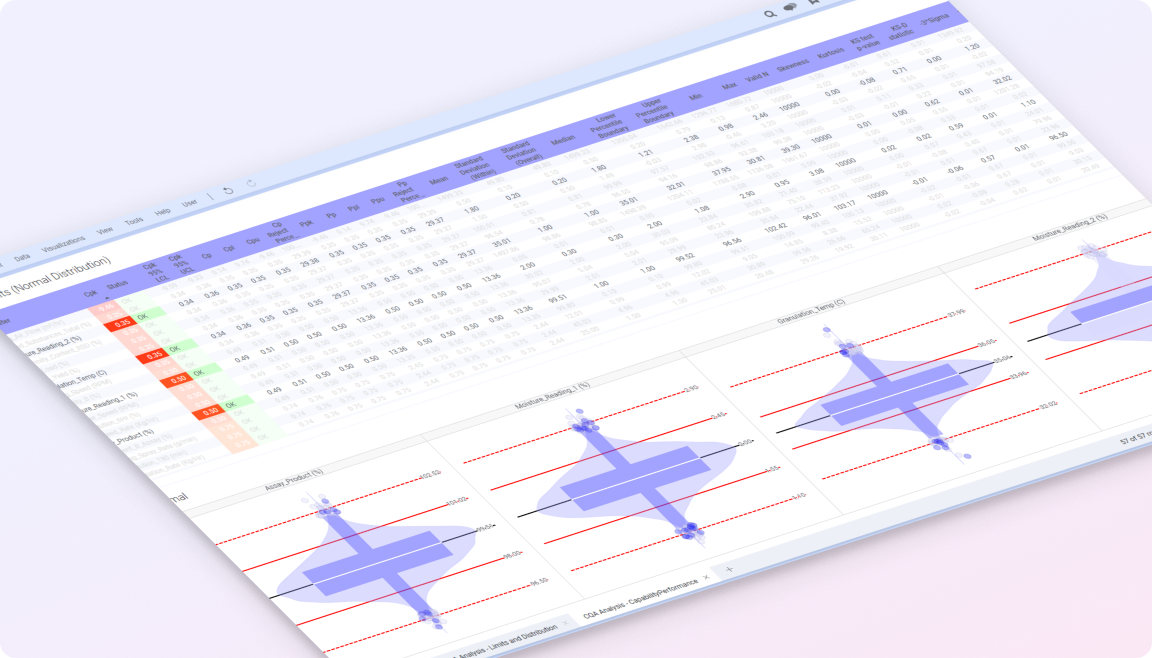
Spotfire brings these elements together in a single visual environment, connecting real-time and historical data with advanced analytics. Teams can monitor variability across batches, investigate deviations in context, and understand how process conditions impact product quality and compliance. The result is more consistent production, faster issue resolution, and greater confidence in meeting regulatory and quality standards.

One decision layer for life sciences manufacturing.
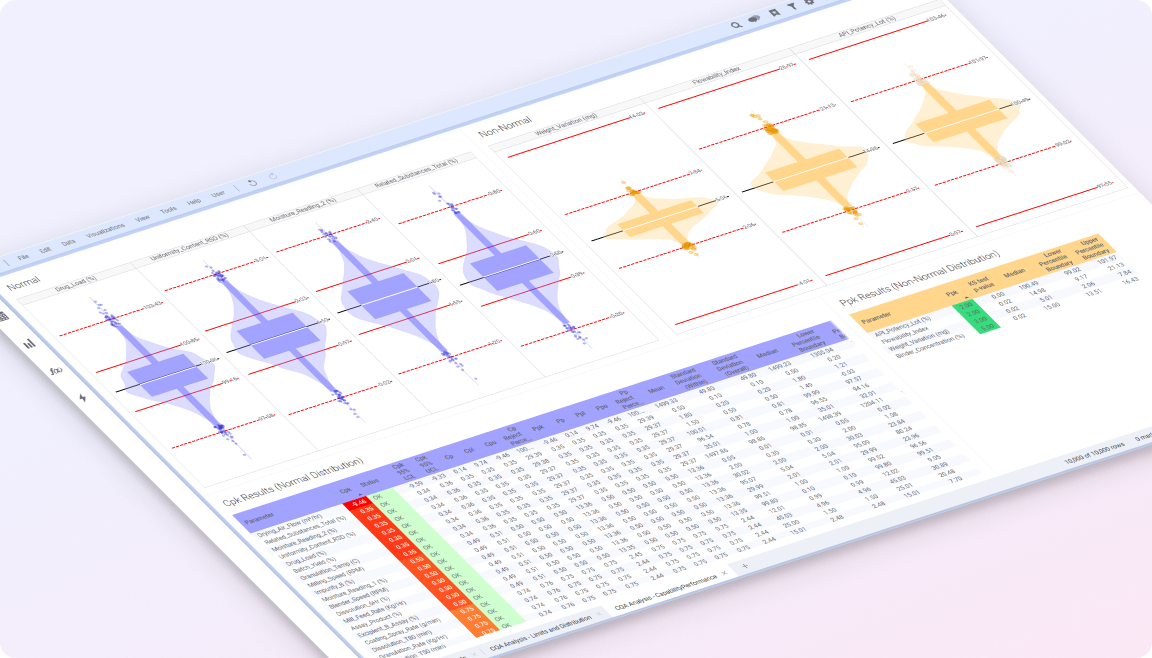
Life sciences manufacturing operates under strict regulatory control, where product quality, process consistency, and traceability are critical. Yet data across production, quality systems, and process monitoring is often fragmented, limiting visibility, and slowing response.
Spotfire brings these perspectives together in a shared analytical environment, enabling teams to understand process behavior, detect deviations earlier, and act with confidence.

From data to decisions in life sciences
Gilead improves drug safety through connected insight
Spotfire enables Gilead to bring fragmented data together, uncover critical insights sooner, and collaborate effectively across teams.
Align teams. Maintain control. Act with confidence.
Life sciences manufacturing decisions span production, quality, engineering, and compliance teams, each working with different data, systems, and perspectives. Spotfire brings these groups together in a shared analytical environment, where they can explore the same data, apply their expertise, and act with alignment.
By connecting batch, process, and quality data in one place, teams gain the visibility needed to detect variability early, investigate issues in context, and maintain control and traceability across operations. In regulated environments where small variations can have significant impact, this ability to understand and respond early is critical. Spotfire helps life sciences organizations discover insights across processes and batches, surface risks before they escalate, and drive operations forward while maintaining quality and compliance.